|
Structural glasses
A glass can be viewed as a liquid in which a huge slowing down of the diffusive motion of the particles has destroyed its ability to flow on experimental timescales. The slowing down is expressed through the relaxation time, that processes relax to equilibrium. Cooling down from the liquid phase, the slow degrees of freedom of the glass-former are no longer accessible and the viscosity of the undercooled melt grows several orders of magnitude in a relatively small temperature interval.
|
As a result, in the cooling process, from some point on, the time effectively spentat a certain temperature is not enough to attain equilibrium: the system is said to have fallen out of equilibrium. The preparation, indeed, plays a fundamental role to get a glass out of a liquid, thus avoiding the crystallization of the substance.
Depending on the material, the ways of obtaining a glass are very diverse and consist not only in the cooling of a liquid but also include compression, intense grinding or irradiation of crystals with heavy particles, decompression of crystals that are stable at high pressure, chemical reactions, polymerization, evaporation of solvents, drying, deposition of chemical vapors, etc..
Many kinds of materials present a glass phase at a given external condition if prepared in the proper way: for instance silica, halide and chalkline based glasses, carbon-based polymer glasses like polyvinylchloride (PVC), germanate dioxide, orthoterphenyl (OTP), open network liquids.....The list is immense and for an exhaustive literature the reader can refer to the books of Donth (2001), de Jong (2002), Rao (2002), Mysen & Richet (2005), Ngai (2011), just to mentiona few.
Without going into details on the huge specificity of glassy compounds, a good glass former can be defined as a system in which noncrystalline packing modes of the molecules are intrinsically at low energy and different modes
are separated by high energy barriers. Apart from rare, often explicitly constructed exceptions, the crystal state is always at lower energy, but the probability of germinating a crystal instead of a glass during the vitrification process is negligible when cooling fast enough: the nucleation of the crystal phase is practically inhibited. In a nucleation event a small but critical number of unit cells of the stable crystal state combine on a given characteristic timescale, the nucleation time nuc. In a good glass former, the number of molecules involved in the nucleation must be much larger than the number of molecules cooperating in the structural relaxation of the glass phase (composing what is called a cooperative rearranging region - CRR), yielding, in this way, a nucleation time much longer than the structural relaxation time. A large nucleation time means that the probability that a fluctuation takes place, allowing a critical number of unit cells to form a crystal, is low. In certain (computer) binary solutions [Kob & Andersen, 1994, 1995a,b; Hansen & Yip, 1995; Parisi, 1997b], at low temperature, the amorphous state is even thermodynamically preferred to any crystalline structure! The same appears to occur for the atactic vinyl polymers, whose lowest energy conformations cannot pack in a regular structure [Gibbs & Di Marzio, 1958].
Many processes are involved at the glass transition, or better, around it, since the transition region depends on the way it is reached in an experiment, and the timescales of the processes play an essential role for the properties and the behavior of the glass former.
Many kinds of materials present a glass phase at a given external condition if prepared in the proper way: for instance silica, halide and chalkline based glasses, carbon-based polymer glasses like polyvinylchloride (PVC), germanate dioxide, orthoterphenyl (OTP), open network liquids.....The list is immense and for an exhaustive literature the reader can refer to the books of Donth (2001), de Jong (2002), Rao (2002), Mysen & Richet (2005), Ngai (2011), just to mentiona few.
Without going into details on the huge specificity of glassy compounds, a good glass former can be defined as a system in which noncrystalline packing modes of the molecules are intrinsically at low energy and different modes
are separated by high energy barriers. Apart from rare, often explicitly constructed exceptions, the crystal state is always at lower energy, but the probability of germinating a crystal instead of a glass during the vitrification process is negligible when cooling fast enough: the nucleation of the crystal phase is practically inhibited. In a nucleation event a small but critical number of unit cells of the stable crystal state combine on a given characteristic timescale, the nucleation time nuc. In a good glass former, the number of molecules involved in the nucleation must be much larger than the number of molecules cooperating in the structural relaxation of the glass phase (composing what is called a cooperative rearranging region - CRR), yielding, in this way, a nucleation time much longer than the structural relaxation time. A large nucleation time means that the probability that a fluctuation takes place, allowing a critical number of unit cells to form a crystal, is low. In certain (computer) binary solutions [Kob & Andersen, 1994, 1995a,b; Hansen & Yip, 1995; Parisi, 1997b], at low temperature, the amorphous state is even thermodynamically preferred to any crystalline structure! The same appears to occur for the atactic vinyl polymers, whose lowest energy conformations cannot pack in a regular structure [Gibbs & Di Marzio, 1958].
Many processes are involved at the glass transition, or better, around it, since the transition region depends on the way it is reached in an experiment, and the timescales of the processes play an essential role for the properties and the behavior of the glass former.
A glass story
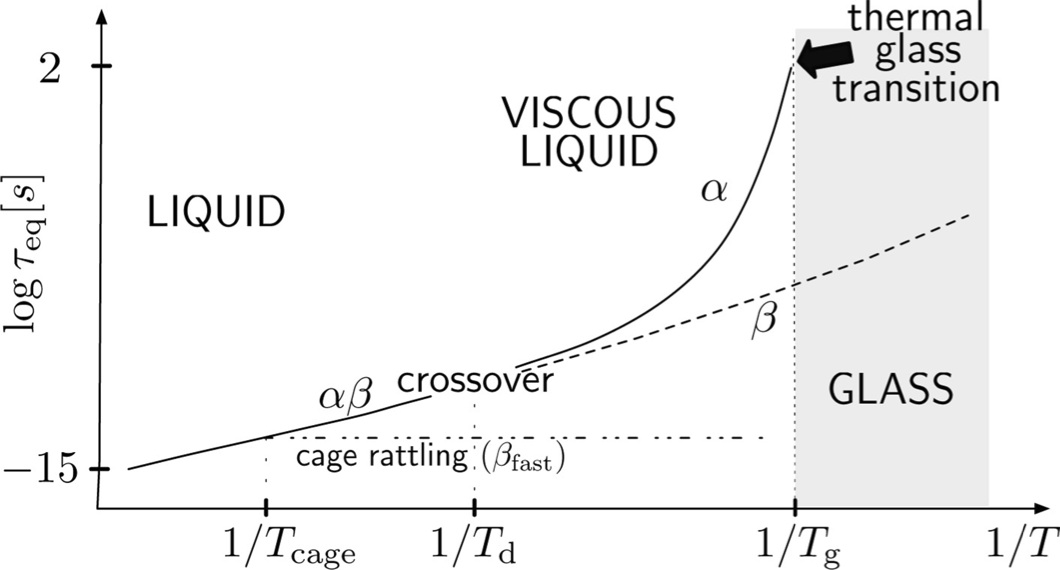
Imagine following a liquid glass former during a cooling procedure, starting from a high temperature. Look at the figure above as a guide, starting from the left side. Already in the warm liquid, different processes occur on different timescales. At a given temperature, that we will simply denote as cage temperature, the thermal movement of particles is slow enough for the diffusion to be hindered by the formation of cages. A cage is, in this case, a dynamic concept relative to each particle in the liquid, whose motion is constrained to occur next to other particles around it, with which it collides like in a three dimensional (floating) pinball machine. This is different than the purely collisional motion taking place in the warm liquid. The timescale of such an inside-cage process is sometimes termed the rattling time. Cooling further, a first bifurcation of timescales takes place between the relative fast rattling time and the relaxation time for the system. The relaxation time is, here, the characteristic
timescale of the process of diffusion from the cage, that becomes longer and longer as the temperature decreases. This process is named in many different ways in literature. Staying close to the notation of Donth we choose the name alphabeta . In summary, the relaxation time in this temperature region is the characteristic
time needed to have one long distance diffusion process of a particle while it is rattling with a high frequency among its neighbor particles forming a (dynamic) cage around it.
timescale of the process of diffusion from the cage, that becomes longer and longer as the temperature decreases. This process is named in many different ways in literature. Staying close to the notation of Donth we choose the name alphabeta . In summary, the relaxation time in this temperature region is the characteristic
time needed to have one long distance diffusion process of a particle while it is rattling with a high frequency among its neighbor particles forming a (dynamic) cage around it.
Dynamical glass transition
Cooling further, in the so called crossover region (always refer to the figure), a second bifurcation of timescales takes place between processes involving a global rearrangement of the system, thanks to the large cooperativeness of the particles (we will call them processes), and processes that involve only a limited number of molecules in a local, microscopically small, rearrangement, thus not contributing to the structural relaxation of the glass former. The latter are usually called processes beta. We reserve the label alpha for the slowest processes, needing a huge cooperativeness to occur below the crossover region, stressing that, in general, different molecular mechanisms may be responsible for the lower temperature processes and the higher temperature
processes. In the crossover region, thus, processes bifurcate in processes - whose characteristic timescale is the structural relaxation time - and processes with a much shorter characteristic time: this way, we are in the
presence of a separation of timescales, that becomes more and more enhanced as temperature is lowered.
Cooling further, in the so called crossover region (always refer to the figure), a second bifurcation of timescales takes place between processes involving a global rearrangement of the system, thanks to the large cooperativeness of the particles (we will call them processes), and processes that involve only a limited number of molecules in a local, microscopically small, rearrangement, thus not contributing to the structural relaxation of the glass former. The latter are usually called processes beta. We reserve the label alpha for the slowest processes, needing a huge cooperativeness to occur below the crossover region, stressing that, in general, different molecular mechanisms may be responsible for the lower temperature processes and the higher temperature
processes. In the crossover region, thus, processes bifurcate in processes - whose characteristic timescale is the structural relaxation time - and processes with a much shorter characteristic time: this way, we are in the
presence of a separation of timescales, that becomes more and more enhanced as temperature is lowered.